Offering more quality transparency in digital health applications for citizens, so that good apps can be used in healthcare: With this goal in mind, we are developing an online app search with our Weisse Liste colleagues. In this context, we are developing the AppQ core-set of quality criteria, funded by the German Federal Ministry of Health. The core-set aims to help standardize the quality requirements for health apps and to provide structured quality data for these apps (as previously reported). In this second blog post on AppQ, we report on the progress of our project and present the draft of the core-set of quality criteria.
The starting point for the development of the AppQ core-set of quality criteria is the meta-criteria catalogue AppKri developed by Fraunhofer FOKUS. The first step of our project consisted of extracting crucial criteria from AppKri that are suitable for describing the quality of a health app regardless of its type or medical topic. In the initial project phase, we created a working draft (core-set 0.9) for this purpose, which we present below:
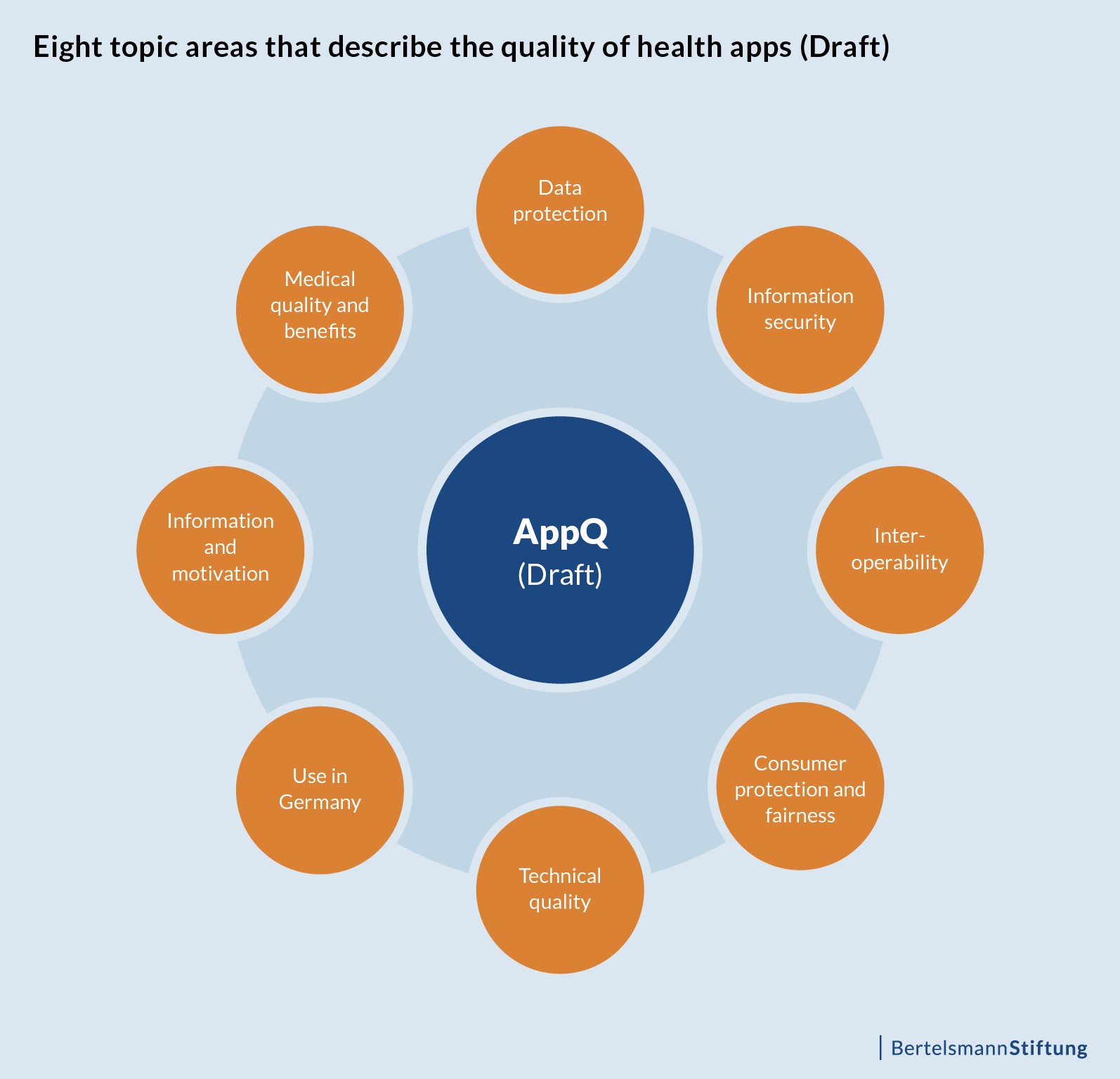
Eight topic areas that describe the quality of health apps
Data protection: Under this heading, relevant criteria cover how regulatory data protection requirements are implemented, whether the user’s consent is requested in a data protection-friendly manner, whether the privacy of the user is protected and whether the requirements of data economy are upheld.
Information security: Here it is displayed whether the provider of a health app can guarantee data security and whether the confidentiality of personal data is guaranteed by the use of current encryption technologies.
Interoperability: Criteria for interoperability include the possibility for users to export data, the availability of standardised interfaces and options for interaction with other digital applications.
Consumer protection and fairness: This topic includes the provision of conclusive app information in the App Store and the consumer-friendly design of the terms of use.
Technical quality: This topic includes criteria for quality control, for the quality of medical-technical functions, as well as for robustness against malfunctions and operating errors of an app.
Use in Germany: The aim here is to identify the extent to which a health app can be integrated into the German healthcare system, for example through interaction with (future) eHealth services of the telematics infrastructure.
Information and motivation: This topic area covers whether a health app is easy and intuitive to use as well as customizable, whether it contains motivational elements for the user, whether instructional measures can be implemented in everyday life and whether the user is supported with appropriate health information.
Medical quality and benefits: This section includes criteria for the medical knowledge base of an application, patient safety, medical efficacy and benefits of a health app for the intended user group.
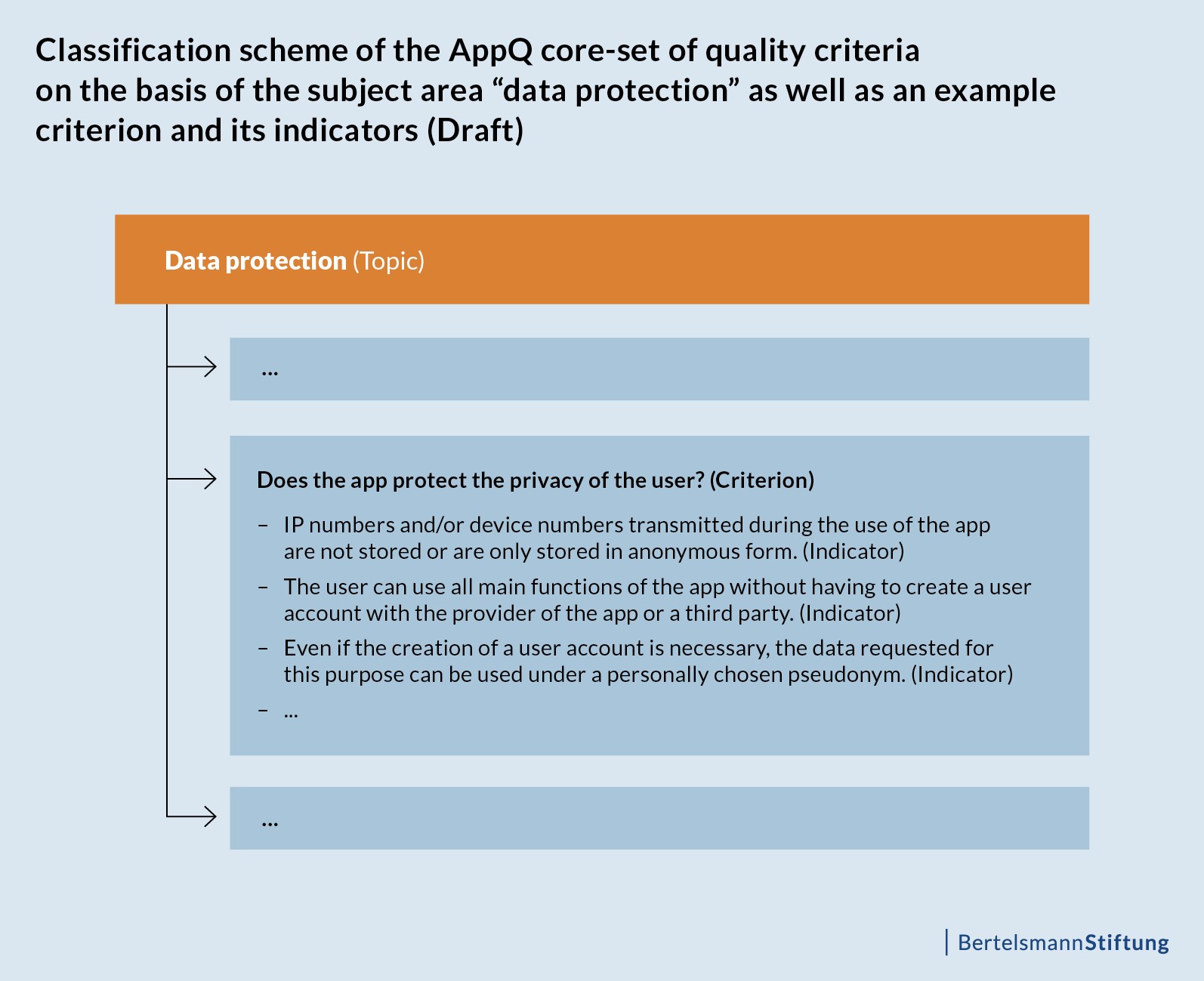
AppQ core-set: topics, quality criteria and their indicators
The quality topics listed describe the draft of the core-set at “heading level”. Each of the eight topics comprises up to six quality criteria, which are phrased as questions and are used to specify and refine the respective topic. The individual quality criteria are supplemented by indicators in order to operationalise the core-set. The following figure illustrates the classification scheme of AppQ using an example:
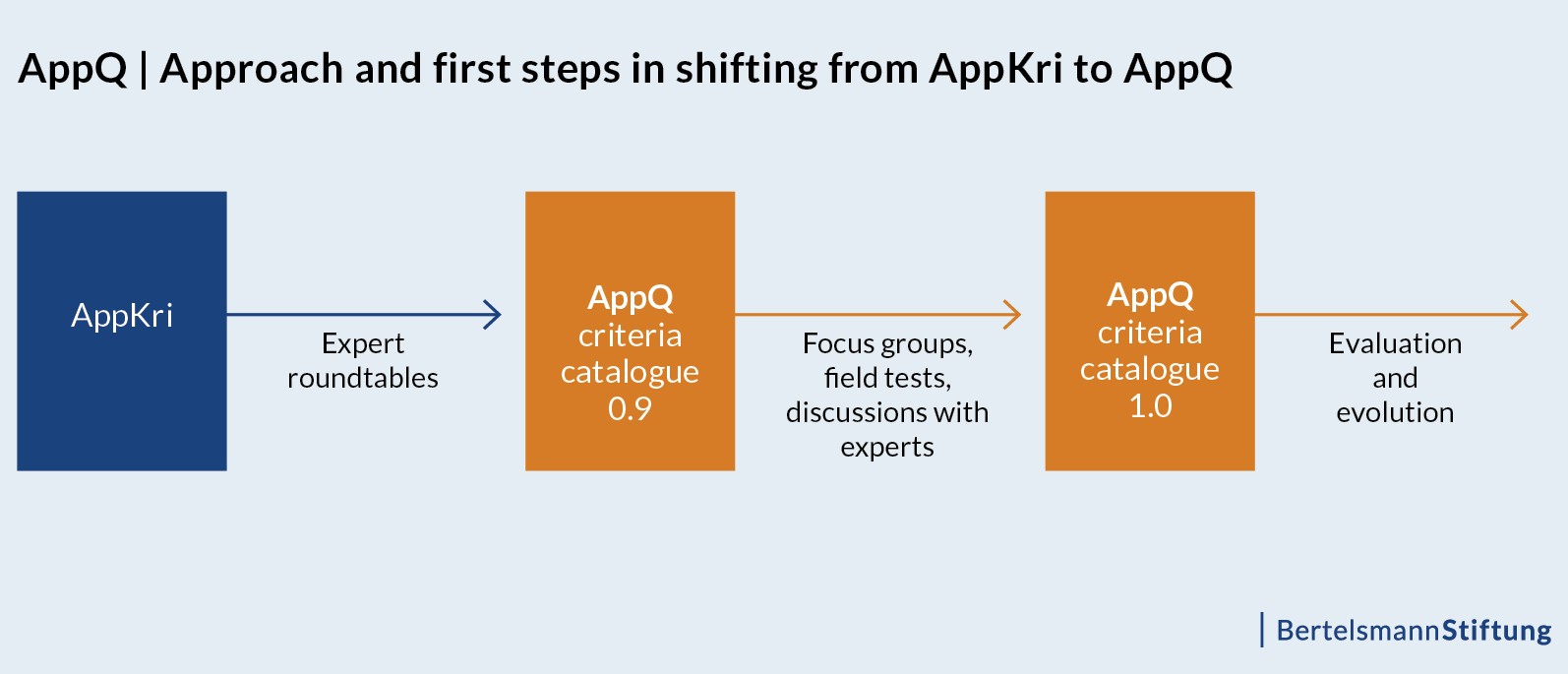
From core-set 0.9 to core-set 1.0
In the next step, the working draft – the AppQ Core-Set 0.9 – will be validated by qualitative focus groups with patients and physicians, as well as by expert discussions and field tests before it will be finalized (see figure below).
As outlined in the previous blog post, AppQ goes beyond the definition of the core-set of quality criteria: During the second project phase, we will make the quality criteria available in a web application so that providers of health apps can present the quality of their applications in a structured manner and make it visible to potential users. Third parties, such as professional medical associations, will also be able to access the quality data of the providers in order to check and evaluate apps belonging to their indication area.

We’ll be providing updates in this blog on the progress made with AppQ. If you’re interested in staying up-to-date with new blog posts, we recommend that you sign up to our newsletter. Please feel free to submit questions or suggestions regarding the project by either submitting a comment here or sending us an e-mail.